Names of deceased
The deceased are Eugenia Atsupui Gakper – 44 years, Alhaji Yobi Banda – 78, Kwadwo Agyei Kyeremeh – 31, and Debrah Leone – 42 years.
Police detain 2 health workers
Police have detained two health officials – 65-year-old James Yeboah, a retired field technician at the Disease Control Unit, who administered the injection; and Simon Tarkrama, aged 42, a physician assistant in charge of the New Senchi Health Centre – who are assisting with investigations.
Health worker was operating illegally – GHS
According to Ghana Health Service (GHS), the man, who has been identified as James Yeboah, had not been authorised to offer such healthcare and had been ordered to stop practising.
Director of Public Health at the Ghana Health Service, Dr Badu Sarkodie said, “Indeed, James Yeboah has been stopped from practising as he was not authorised by the Ghana Health Service at the national, regional or district level to conduct this activity in the facility. So he has been stopped from practising in that facility to make sure that he doesn’t put other people at risk.”
Adverse reactions after injection
Adverse reactions described as injection site abscess, skin necrosis and ulcers were reported during the third week of March 2018 to the FDA when the patients had injections of Benzathine Penicillin at the health centre in the Asuogyaman District.
Initial police investigations
Initial police investigations said on March 29, 2018, at 4pm, Asuogyaman District Director of Health, Abdul Aziz Abdulai brought Simon Tarkrama and James Yeboah to the police station to file a complaint.
Abdulai told the police that on March 19, 2018, he received a phone call from the medical superintendent of Volta River Authority (VRA) Hospital, Akosombo, that three patients were admitted at the hospital after they received injection from New Senchi Health Centre.
Eugenia Atsupui Gakper, 44 years
Eugenia Atsupui Gakper died on March 25, 2018 at VRA Hospital. She passed away days after she took injections to treat some skin infections.
She took three injections in the buttocks on March 9, 12 and 13, and complained of severe pains thereafter.
Alhaji Yobi Banda, 78 years
Alhaji Yobi Banda also died on March 27, 2018 at VRA Hospital.
Kwadwo Agyei Kyeremeh, 31 years
On March 28, 2018, Kwadwo Agyei Kyeremeh, who was referred to the Korle-Bu Teaching Hospital, also died there.
Kwadwo Adjei Kyeremeh, 31, was the administrator of the Rev Friedrich Monninger Memorial Presbyterian Church in Akosombo.
Kyeremeh and Eugenia preparing for marriage
Both Kyeremeh and Eugenia were in the same congregation and they were both preparing for their separate marriages this year.
Debrah Leone, 42 years
The fourth victim, Debrah Leone, who was referred to the Volta Regional Hospital at Ho, also died on Saturday, March 31, 2018.
Regional medical investigators at work
On March 30, 2018, a team of medical investigators from the Regional Health Directorate in Koforidua extended enquiries to VRA Hospital at Akosombo, where they met Dr Nartey, a medical officer at the facility.
VRA Hospital at Akosombo
According to Dr Nartey, the medical superintendent at VRA Hospital was not around to give a report or statement on the matter.
He, however, led police to the hospital mortuary, where deceased Kwadwo Agyei Kyeremeh and Eugenia Gakper were seen in the morgue.
Akuse Government Hospital
According to the police, further enquiries were extended to Akuse Government Hospital, where it was alleged that victim/deceased Alhaji Yobi Banda was sent for treatment.
The preliminary report said medical personnel the team met on duty at the hospital told police that the hospital administrator was not at post, hence could not comment on the issue.
Police say efforts are being made to get a report from the medical superintendents at VRA and Akuse as soon as possible.
FDA pharmacovigilance team investigates
Meanwhile, the Food and Drugs Authority (FDA) has said a pharmacovigilance team from the Eastern Regional office has been dispatched to the New Senchi Health Centre to investigate injection reaction reports which led to the death of four patients.
It said serious adverse reactions described as injection site abscess, skin necrosis and ulcers were reported during the third week of March 2018 to the FDA when the patients had injections of Benzathine Penicillin at the health centre in the Asuogyaman District.
It said the investigation would be undertaken in collaboration with the Ghana Health Service and would keep the public updated as more information becomes available.
A statement signed by FDA Chief Executive Officer, Mrs Delese Mimi Darko, said preliminary investigations revealed that those reactions may be due to contaminated 0.9 per cent Normal Saline, the solution which was used to reconstitute (mix) the Benzathine Penicillin Powder for injection.
Normal Saline Solution used repeatedly for several days
The 0.9 per cent Normal Saline Solution was reported to have been opened and used repeatedly for several days, which might have resulted in the contamination.
The statement said the Benzathine Penicillin was presented as powder for injection and should be reconstituted with sterile water for injection and also used immediately to avoid contamination.
Meanwhile, healthcare professionals and consumers have been encouraged to be vigilant and report untoward effects of medicines and health products to the FDA.
They could report by completing adverse reaction reporting forms provided in hospitals and community pharmacies designated as Patient Safety Centres across the country.
Injection death toll rises
- Posted on
- Comment
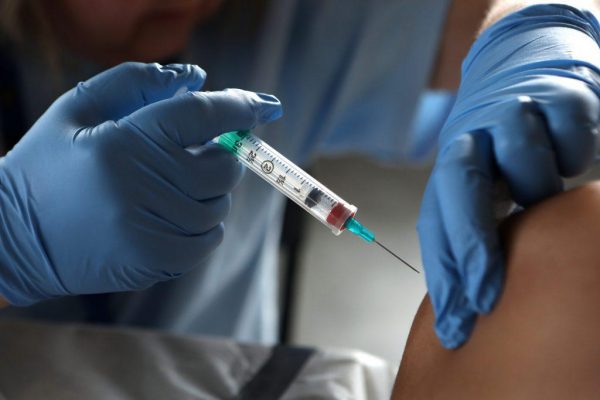 -Ghana Health Service says health worker was operating illegally
-Ghana Health Service says health worker was operating illegally
The number of people who have died from injections received at the New Senchi Health Centre at Akrade in the Asuogyaman District of the Eastern Region has risen to four.
By Elvis DARKO, Accra






 (Selorm) |
(Selorm) |  (Nana Kwesi)
(Nana Kwesi)